About
About this poster:
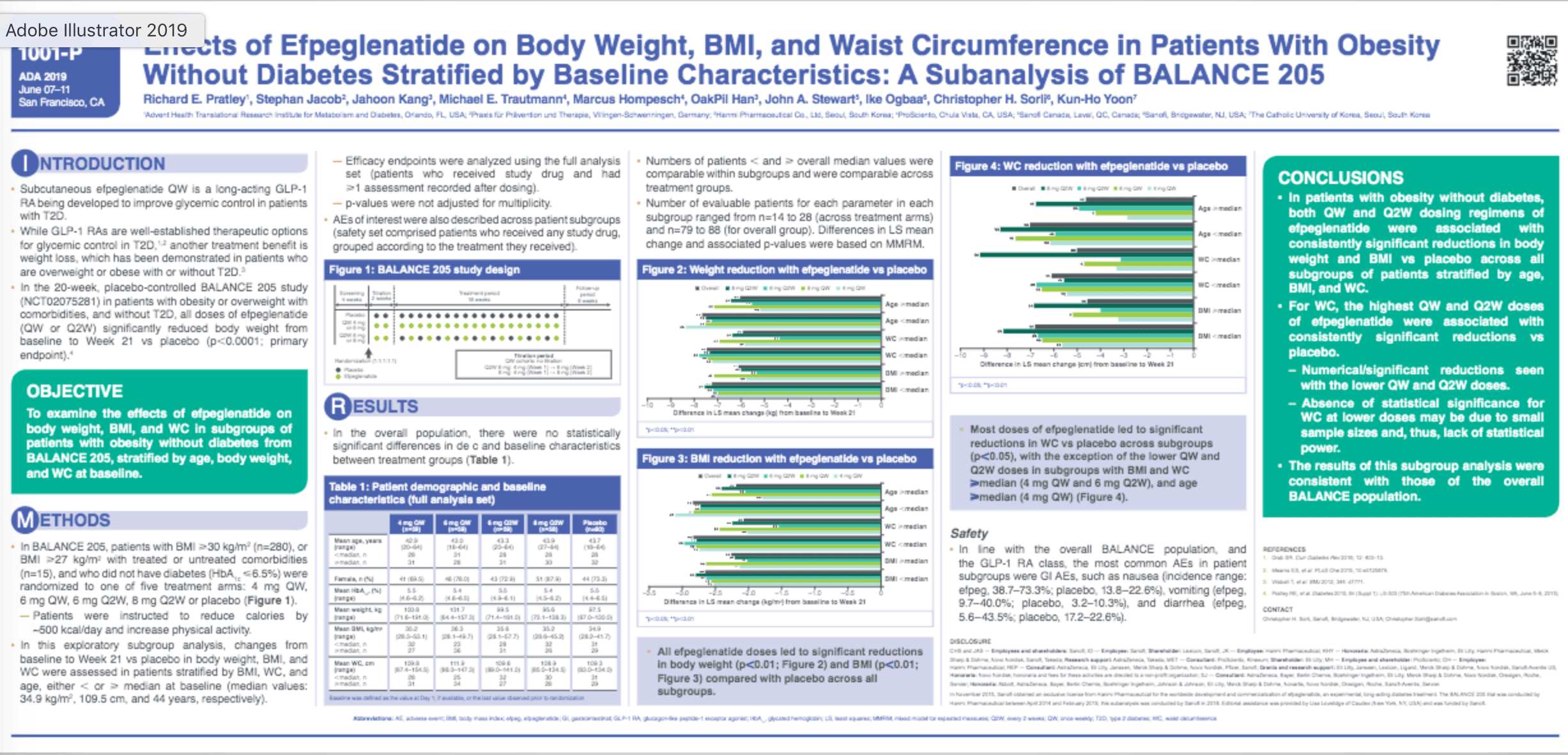
Once-weekly (QW) subcutaneous efpeglenatide (efpeg) is a long-acting GLP-1 RA being developed to improve glycemic control in patients with T2D. In the BALANCE 205 study (NCT02075281) in patients with obesity (median BMI 34.9 [28.1−57.7] kg/m2) without diabetes, all doses of efpeg (4 or 6 mg QW; 6 or 8 mg once every 2 weeks [Q2W]) significantly reduced body weight (BW) from baseline (BL) to Week 21 vs. placebo (PBO, p<0.0001; primary endpoint). In this exploratory subgroup analysis, the effects of efpeg (in each treatment arm or pooled data) on BW, BMI, and waist circumference (WC) from BL to Week 21 vs. PBO were investigated in patients stratified by BMI, WC, and age (< or ≥ median BL values). All efpeg groups were associated with significant reductions in BW and BMI from BL vs. PBO across all subgroups (Table). Efpeg also led to significant reductions in WC vs. PBO across subgroups apart from lower doses of efpeg in subgroups with BMI and WC ≥ median (4 mg QW and 6 mg Q2W), and age ≥ median (4 mg QW). The observed adverse events mirrored those known for GLP-1 RAs.
In conclusion, all doses of efpeg led to consistent, significant reductions in BW and BMI in patients with obesity without diabetes in all subpopulations tested; results were consistent with those of the overall BALANCE population.